CALCIUM
Written by Slawomir (“Swavak”) Gromadzki, MPH

Calcium is a mineral and alkaline earth metal. It serves a number of physiological functions. Its ions within the body are necessary to send signals to-and-from the brain. One of its most apparent functions is that of maintaining firm structure of bones and teeth. More than 90% of body calcium is stored in these two structures. Calcium is also one of the main components of hair, and nails. Only 1 percent of calcium is stored in other body tissues, which means we need to avoid calcium accumulating in other parts of the body.
Calcium is needed to control levels of magnesium, potassium and phosphorus in the blood, and all these minerals work together to balance one another. This is one of the reasons why it is so important to avoid excess of calcium and at the same time consume mineral- and calcium-rich foods on a regular basis as it helps avoid electrolyte imbalance.
DEFICIENCY
I am not 100% sure it is true, but the World Health Organization has stated that “calcium deficiency has never been documented anywhere in the world, even with calcium intakes of as little as 300 mg/day.” There is a reason to believe that calcium deficiency is a problem in spite of high dairy consumption. However, it is not caused by insufficient intake of this mineral but it is a result of very common vitamin D deficiency as well as high intake of animal protein, sugar and caffeine. Also the fact that soil is now much lower in calcium can in some degree contribute to the problem as calcium levels in food are declining too.
Nevertheless, whether this surprising WHO statement is right or not, the truth is it is not calcium deficiency which should concern people so much but rather its absorption in our body and excessive intake of calcium in dairy and in supplemental forms. Also in terms of bone health, it isn’t the lack of dietary calcium that causes osteoporosis, but rather eating too much animal protein, especially in the form of milk and dairy products (see evidence below), deficiency of vitamin K2 and D, as well sedentary lifestyle.
Most common calcium deficiency symptoms may include the following:
- Brittle and weak bones
- Muscle spasms and cramps
- Problems with blood clotting
- Tiredness
- Weakness
- Feeling pins or needles
- Irritability
- Delayed growth and development in children
- Irregular heart beat
- Blood pressure problems
HEALTH BENEFITS
MUSCLES
Calcium prevents muscles from being overactive and going into spasms. The truth is, however, calcium deficiency rarely contributes to muscle cramps. In most cases it is rather due to magnesium deficiency and some other factors. Excess calcium may even cause muscles to contract too much, and lead to the deficiency of magnesium which is needed to relax muscles. Vitamin D is very important, but when you take it with a calcium supplement and without enough magnesium, it is easy to get a calcium overload that can cause cramps, or make existing cramps worse.
Testimonial: “When I was in my mid-twenties I had suffered from menstrual cramping, and someone recommended taking calcium lactate (capsules-not tablets). It worked, and I never had menstrual cramps again. I’m now passing my mid-fifties, and over this past summer ‘was getting leg cramps in the middle of deep sleep. I purchased and began taking a calcium complex, and got relief right away. I usually only need to take half the dose, which is: 200 IU Vit. D3, 500 mg Calcium, 250 mg Phosphorus, and 250 mg Magnesium. Whenever I forget to take them, the cramps come back the same as before. My husband is also getting relief of leg pain now taking this dose”. (Source >)
Natural organic calcium found in calcium-rich plant foods and also in the form of some good supplements, such as calcium citrate, can help reduce heartburn symptoms because natural organic calcium helps the lower oesophageal muscle sphincter valve that controls food moving into the stomach to close properly. Unfortunately, deficiency of calcium and especially magnesium leads to malfunctioning of this muscle allowing acid from stomach to enter eosophagus causing burning sensation and acid reflux. Taking calcium and magnesium improves this muscle function and can often help reverse the symptoms associated with acid reflux >.
On the other hand, the inorganic calcium carbonate (often used as one of the ingredients in antacids), although helps lower stomach acid production thus reducing heartburn, yet in reality this effect causes more harm making the problem worse. It is so because about 80 percent of acid reflux cases are caused by low stomach acid as it leads to the malfunction of the lower oesophageal sphincter. In order for the sphincter to close properly sufficient amount of stomach acid is required. In addition, the reflux is also caused by food that has been staying in the stomach due to deficiency of acid. It ferments, creating gas and pressure that reopens the sphincter valve between esophagus and the stomach allowing acid to travel up into eosophagus.
BLOOD CLOTTING
Like deficiency of vitamin K (a common problem today), low calcium in the blood can also lead to the inability of the blood to clot.
NERVE IMPULSES
Convulsive seizures and inability to transmit nerve impulses may be caused by calcium deficiency. Calcium helps release neurotransmitters in the brain that control nerve signalling and muscle movements. It activates certain body proteins that are needed by muscles to move and contract. Calcium also helps with the control and release of glucose (sugar) in the bloodstream, which is the key fuel used by the brain cells and muscles.
IRREGULAR HEARTBEAT
Since calcium is required for proper nerve transmission, it plays an important role in regulation of the heartbeat. Its deficiency, therefore, may lead to irregular heartbeat.
BONE HEALTH
Calcium increases bone density and also reduces the prevalence of osteoporosis. About 80% of people with osteoporotic fractures had insufficient daily intake of calcium and especially vitamin K2 and vitamin D.
However, it is not the insufficient intake of calcium but rather regular consumption of dairy products (high animal protein intake) that leads to bone calcium loss and osteoporosis. To prevent and treat osteoporosis, it is therefore much more important to reduce or eliminate dairy from the diet and take good quality supplements with vitamin K2 (MK7), vitamin D3 and magnesium. Magnesium is needed here not only to strengthen bones but also because if you are deficient in it vitamin D will not give any benefit.
PMS
Calcium can be useful in relieving PMS symptoms, including cramps, headaches, mood swings, breast tenderness, muscle aches, and fatigue (>). During menstrual cycle estrogen levels increase causing calcium concentrations to drop. For this reason taking a good quality calcium supplement, such as calcium citrate (preferably 100% powder form) may help to balance this relationship and reduce symptoms.
CANCER
There is a significant association between calcium and vitamin D intake and a reduced risk of death from at least 15 types of cancers (>). Calcium is important in a process called apoptosis, which leads to the death of abnormal cells in the body, thus preventing them from causing cancer. Every time a cell divides, it is supposed to pause and check the integrity of its DNA. If the DNA is damaged, it tries to repair it. And if it can’t be repaired the apoptosis is triggered and the damaged (abnormal) cell dies.
300 mg of calcium intake per day was associated with an approximately 8% reduced risk of colon cancer (>). Malfunction of APC/β-catenin pathway is a common problem which is associated with increased risk of colon cancer. Calcium supplements have shown to improve the pathway and reduce the risk (>).
BLOOD PRESSURE
Healthy calcium intake may lower the probability of someone having high blood pressure. Calcium is involved in supporting normal heart function and healthy blood pressure because of its role in dilating blood vessels and sending chemical nerve signalls from the brain to the heart. Supplemental dietary calcium has been shown to help lower high blood pressure, whereas lower calcium levels tend to elevate blood pressure (>).
WEIGHT MANAGEMENT
Calcium may help lose weight which benefit may be explained by improving appetite control and an increase in fat oxidation (>). This effect is more pronounced when calcium is combined with supplemental vitamin D intake. Together they increase whole body fat oxidation after meals, increase fat excretion and decrease hunger (>).
DIABETES
Calcium and Vitamin D effect pancreatic cells that control insulin secretion and therefore promote healthy blood sugar levels.
BEST DIETARY SOURCES OF CALCIUM
Dairy products (milk, cheese, yogurt, etc.) are always mentioned as the best source of calcium. Unfortunately, due to high content of animal protein, which causes bone loss, the healthiest source of this mineral is not dairy but vegetables (especially leafy greens), sesame seeds, legumes (especially organic soya products such as tofu) and grains such as amaranth:
Lambsquarters (edible weed) (1 cup = 450 mg)
Collard greens (1 cup = 350 mg)
Amaranth grain (1 cup = 300 mg)
Figs, dried (10 = 270 mg)
Tofu, firm (½ cup = 260 mg)
Turnip greens (1 cup = 250 mg)
Sesame seeds, dried (2 tbsp = 180 mg)
Kale (1 cup = 180 mg)
Blackstrap molasses (1 tbsp = 170 mg)
Okra, boiled (1 cup = 150 mg)
Baked beans (1 cup = 150 mg)
Artichoke, boiled (1 medium = 135 mg)
Quinoa grain (1 cup = 100 mg)
THE HIGHER DIETARY CALCIUM INTAKE THE BETTER?
Perhaps the dietary nutrient that still attracts more attention than any other is calcium, mainly due to the influence of the dairy industry trying to promote its produce (high in calcium).
Because excess calcium is harmful, the body has a mechanism that prevents the over absorption of calcium. As a result only 20-30% of this mineral ingested in food is absorbed. It is one of the reasons we shouldn’t take calcium supplements or consume foods, such as dairy, that are too high in calcium.
According to certain interesting experiment, two groups of boys showed a significantly different ability to absorb calcium. Participants from the group on a high calcium diet were able to retain only about 100 milligrams of calcium per day. On the other hand, the other group fed on a low calcium diet for two months were able to take up almost 400 milligrams of calcium per day. The group on high calcium diet failed to assimilate calcium from food and simply excreted (lost) most of this mineral.
The loss of calcium is not only the result of its excessive intake but it is caused especially by high intake of animal protein found in dairy. A high protein, low carbohydrate diet triggers a mechanism for loss of calcium from bones.
A good indicator proving that humans need much less calcium and protein than the amounts found in cow milk and dairy is human breast milk content. It contains only 80 milligrams calcium per cup, whereas cow’s milk contains almost 300 milligrams per cup. The difference between protein content in these two sources is even greater as human breast milk is very low in protein compared to cow’s milk. The difference is related to the fact that human babies grow very slowly and do not need as much protein as fast growing calves. That is why cow’s milk and dairy are not fit for human consumption (including adults).
The American Pediatric Association has stated that new born babies and infants below two years of age should not be fed with cow’s milk due in part to the baby’s leaky gut that allows a passage of unhealthy animal constituents to enter their blood stream, causing allergic reactions, ear infections, sinusitis and other problems.

HARMFUL EFFECT OF DAIRY ON BONES
Many decades ago researchers found that it is not a lack of calcium that causes health problem (including poor bone health) but rather the high consumption of animal protein. In the effort to digest animal protein, our body produces sulfuric acid, thus creating a very acidic environment, and in order to lower this high acidity the body uses calcium stored in bones thus weakening them. This taken from bones calcium is removed from the body together with acid in urine.
Dr. McDougall blames the dairy industry’s propaganda for brainwashing people and creating “obsession over calcium that has become, in effect, a major contributor to the suffering and death of more than one billion people annually on Planet Earth from diseases of overnutrition – obesity, heart disease, stroke, arthritis, and diabetes”. He also states that, “because of their financial power and political connections, the people representing dairy industry can say whatever they want and no one can stop them”. This is why governments as well as the mainstream media still keep repeating the lie according to which milk is essential to good health while at the same time they ignore the numerous medical studies which repeatedly proved that milk is a serious health threat. In many countries people seem to be easily fooled by the dairy lobby.
The best example can be Americans who consume 3 times more dairy products than grains, and 5 times more than fruits! That is one of the main reasons why there are so many sick and obese people living in America and why the prevalence of osteoporosis and rates of hip fractures are highest in countries such as USA, Sweden, Norway, New Zealand or Australia, where animal protein consumption (including dairy products) is very high. On the other hand, people living in rural regions of Asia or Africa whose diet is based on unrefined plant foods and the consumption of animal protein is low have the lowest rates of osteoporosis and hip fractures. Unrefined plant foods are packed with organic calcium and at the same time contain much lower (but sufficient) amounts of protein, thus having only positive effects on our bones.
For example, the African Bantu women consume no dairy products and their daily calcium intake, which comes from plant sources, is only about 300 mg and even the oldest women among them are free of osteoporosis, as well as many other degenerative diseases. Research has shown that one of the main causes they don’t suffer from osteoporosis is their plant-based and low in protein diet.
On the other hand, the rates of osteoporosis-related bone fractures among Inuit (Eskimo), whose diet is very high in animal protein, are one of the highest of all population groups. It is also interesting that although their diet contains about 2,000 mg of calcium per day (mostly from fish bones) it doesn’t seem to help at all. There is no evidence that a higher calcium intake in adulthood will prevent or reverse osteoporosis.
The highly acidic nature of animal protein such as dairy and meat has been proved to be the major cause of osteoporosis and bone loss. Unlike plant high protein foods, such as legumes (pulses), the animal protein is rich in sulphur which creates a lot of acid during digestion process, thus leading to acidosis. As a result our body tries to lower the acidity and regulate the pH using calcium phosphate. The problem is that this process contributes to osteoporosis as the calcium is taken from the bones thus making them weaker. It means that by consuming milk and dairy products, people are losing more calcium than they actually ingest, thus leading to a negative calcium balance. It is estimated that because of this a stunning 25 million people only in America have been diagnosed with osteoporosis! Yet majority of physicians and nutritionists still encourage us to consume dairy in order to strengthen our bones!
The following very interesting statement by Professor Walter Veith (>) proves milk and animal protein is the true cause of the weak bone problem: “It is true that dairy products do contain large amounts of calcium, but a large proportion of the calcium in milk is combined with casein (protein) in the form of calcium caseinate. Grains, legumes, seeds, nuts and many vegetables are excellent and better sources of calcium because absorption of calcium from these sources is higher than from milk. A study done on whole-wheat products showed that fractional calcium absorption from whole-wheat bread exceeded the absorption of calcium from milk, ingested at a comparable load, in the same subjects. Not only is the absorption rate of calcium higher from plant sources, but animal sources seem to cause calcium loss in view of the nature and the concentration of the proteins which they contain. In addition to the foods listed above, almonds, sesame seeds, sunflower seeds, soybean products such as bean curd and soy flower, carob, chick peas, haricot beans, mung beans, garlic, parsley, watercress and dried fruits (especially figs) are good sources of calcium. Calcium utilization is governed by complex processes, and the absorption, bone deposition and excretion of calcium is strongly influenced by other dietary components. It is well documented that increased protein consumption causes calcium loss in the urine, and this is particularly true in the case of proteins from animal products. An investigation into the effects of various protein diets on calcium retention showed that proteins from dairy products, such as cottage cheese, caused considerable calcium loss in the urine. The culprit here seems to be casein, and other studies seem to corroborate this. In one study it was found that casein fed to weaning rats caused kidney calcification, an effect which was not observed if the protein came from another source. If calcium losses exceed absorption, then a negative calcium balance exists, and calcium must be mobilized from bone in order to maintain plasma calcium levels. This loss of calcium from bone can eventually lead to osteoporosis. It is, therefore, not surprising that osteoporosis seems more prevalent in countries where the consumption of dairy products is high… Moreover, calcium supplementation does not provide a solution for the dilemma, as countries, such as the United States, with the highest supplementation rates still have among the highest rates of osteoporosis. Not even in lactating women does calcium supplementation seem to have much of an effect in either white or black women. In one study on White, middle class women it was found that absorption of calcium from the intestines was not increased during supplementation compared to lactating women on low calcium diets. Gambian women normally have a low calcium diet based largely on rice, millet, groundnuts and some fish. This diet provides less than 300 mg/day of calcium which is considerably lower than current recommendations for lactating women. Increasing the calcium level threefold to bring it in line with FAO/WHO recommendations had no discernable effect on breast milk calcium concentrations or on the maternal bone mineral content. This suggests that there was no benefit from increasing the calcium intake during lactation. The women in the supplementation group had consumed a total of 0.26 kg (one quarter to one third of the whole-body calcium content) after 52 weeks, and showed significantly greater urinary calcium output (7% of the dose). As the bone mineral content did not change, the researchers concluded that the rest of the additional calcium was just not absorbed from the intestinal tract. This study underlines the fact that calcium needs can be adequately met by even low calcium diets that contain no dairy products. In our own laboratory we have consistently found that animals (sheep, rats, rabbits and vervet monkeys) fed animal proteins, particularly casein or dairy products showed significant increases in urinary calcium levels, compromised bone status, and in common with the other studies, reduced intestinal absorption.”
The above quote has been taken from the book Diet and Health by Professor Walter J. Veith available through Amazing Discoveries webstore (>).
THE RISK OF CALCIFICATION

Overconsumption of calcium to prevent osteoporosis creates other mineral deficiencies and causes calcification leading to hardening of arteries (caused by calcium and cholesterol deposits), heart attacks and strokes, calcification of pineal gland, osteoarthritis (caused by calcium deposits), or kidney stones. Excessive calcium intake is also believed to contribute to gallstones, hypothyroidism, obesity and type 2 diabetes.
A 2011 study found that people who took calcium supplements regularly had an 86 percent greater risk of having a heart attack due to calcification (hardening) of arteries caused by excess calcium!
A 2010 meta-analysis showed calcium supplements (without coadministered vitamin D) are associated with a 27% increased risk for heart attack. Even when calcium was administered with vitamin D, which helps absorb and utilize calcium, elemental calcium still increased heart attack risk by 24 percent (only supplementation with vitamin K2 can prevent this harmful effect).
A 2008 study found calcium supplements are associated with a greater number of heart attacks in postmenopausal women.
A 2004 study showed that people with excess calcium in their coronary artery and who take statins have a 17-fold higher risk of heart attacks than do those with lower arterial calcium levels.

CALCIUM SUPPLEMENTS
In most cases (especially if you consume mentioned above plant sources of calcium) you don’t need calcium supplements, but if you need them take only good quality organic forms such as calcium citrate. Be sure supplements are not made from calcium carbonate or bicarbonate, bone meal or oyster shells as these are bad inorganic forms high in calcium carbonate. In addition, these so called natural sources of calcium are often contaminated with arsenic, lead, or mercury that accumulate in the bones and shells.
CALCIUM ABSORPTION
Vitamin D is necessary for proper calcium transport and metabolism and a lot of people simply aren’t getting enough of it unless they take at least 2000-5000 IU of vitamin D3 per day with magnesium which is required for vitamin D activation in the body.
Also Magnesium, phosphorous, and vitamin K are vital for calcium absorption and use in the body.
There is also an evidence that calcium metabolism and our ability to absorb calcium declines very substantially if we don’t have enough vitamin C.
NEVER TAKE CALCIUM WITHOUT VITAMIN K2!
Never take calcium supplements without vitamin K2 MK7, D3 and good magnesium, because if you are deficient in these nutrients (and almost all people today are), calcium you get from supplements, hard water or milk and dairy, instead of strengthening your bones will be deposited in pineal gland (leading to its calcification), kidneys (causing kidney stones), joints (osteoarthritis), veins (varicose veins and haemorrhoids), colon (constipation), and arteries (causing their hardening and leading to heart attacks and strokes).
Studies gave strong evidence that higher calcium intake through supplements can significantly increase heart attacks because excess calcium builds up in the blood vessels causing their hardening and contributing to thickening and blockage. A 139% increase in risk was noted in one study (>).
One of the most important activities of vitamin K2 is to protect the entire cardiovascular system by preventing calcification of the arteries. Please keep in mind that only vitamin K2 and not K1 has this ability. In one study Vitamin K2 reduced blood vessel calcification whereas vitamin K1 did not.
In our blood, Vitamin K2 participates in carboxylation of Matrix Gla Protein (MGP), the most potent inhibitor of arterial calcification known. In this way Vitamin K2 prevents vascular damage by lowering the risk of calcification (hardening) of arteries. This effect is more beneficial when Vitamin K2 is combined with Vitamin D3 and Magnesium. Vitamin K and vitamin D work to increase a substance called Matrix GLA protein (MGP). This protein protects the blood vessels from calcification by congregating around the elastic fibres of the arterial lining and guarding against calcium crystal deposition. Some experts maintain that MGP is the most powerful inhibitor of soft tissue calcification currently known.
Animal studies have shown that vitamin K2 not only prevents hardening of blood vessels but actually has the potential to reverse arterial calcification by activating MGP. A trial followed 16,000 people for 10 years found that each additional 10 mcg of K2 in the diet reduces cardiac events by 9 percent!
The 2004 Rotterdam Study revealed that people who consume 45 mcg of Vitamin K2 per day live seven years longer than people getting only 12 mcg per day. It also demonstrated that they had a 50% lower risk of death from heart disease than people with the lowest rates of K2.
TOTAL VS ELEMENTAL CALCIUM
When purchasing calcium supplements, look for the elemental calcium content, not the total content. For instance, a supplement containing 500 mg of calcium carbonate provides 200 mg of elemental calcium. In calcium carbonate calcium combines with carbon and oxygen. So, the molecule known as calcium carbonate contains elemental calcium, elemental carbon, and elemental oxygen. When calcium carbonate is ingested, the calcium that is bound to carbon and oxygen is broken down to its elemental form. This quantity is known as elemental calcium.
The percentage of elemental calcium in calcium carbonate that is absorbable into the blood in our bodies to absorb, is 40 %. It means that for every tablet that contains 1000 mg of calcium carbonate, 400 mg (40 %) of that tablet is elemental calcium and becomes available for absorption into the blood from the digestive system.
Other calcium supplements work the same way. Another common calcium supplement formulation is calcium citrate, which provides 20 % of the elemental calcium. This means that, if you take a 1000 mg tablet of calcium citrate, you will absorb 200 mg of elemental calcium.
On paper, it looks like calcium carbonate is much better than citrate but in reality the opposite is true due to the fact that citrate is an organic while carbonate an inorganic form and because the absorption and usability of citrate is much better (citrate is recognisable by body cells as “own” and carbonate as “foreign”).
FORMS OF SUPPLEMENTAL CALCIUM
What most people are not aware of is the fact that our body can only utilize organic calcium efficiently and not the inorganic one. In nature, calcium is found in organic and inorganic forms. It is important to know what the organic and inorganic forms are. Organic calcium is obtained from plant sources like unrefined plant foods and algae. Inorganic calcium is obtained from dairy products and is not utilized by the body. In fact, inorganic calcium is poorly absorbed by the body.
Organic calcium ingredients, such as calcium citrate, calcium lactate and calcium gluconate, have higher bioavailability and better solubility than inorganic calcium salts. Consequently, organic sources command higher prices and have a larger segment share, according to Frost & Sullivan market research.
Inorganic calcium ingredients, including calcium carbonate and calcium phosphate, have a high calcium content, but are not as bioavailable as organic options. Most Calcium carbonate is excreted out of urine and not absorbed by the body. Most commonly found calcium supplements are obtained from rocks and available in the form of calcium carbonate. Calcium phosphate and calcium sulphate are also inorganic forms.
Calcium orotate, calcium aspartate, calcium ascorbate are organic forms of calcium with high absorption rate. But these are difficult to find and are expensive.

CALCIUM CITRATE
Calcium citrate is the calcium salt of citric acid. Calcium is best absorbed in an acidic environment, so calcium citrate (a chelate) is the best absorbed supplemental form of calcium as it does not require extra stomach acid for absorption, and you can take it at any time during the day, even on an empty stomach.
Calcium citrate usually provides less elemental calcium per dose than calcium carbonate, but it is organic and much more usable. 500 mg calcium citrate supplement provides 105 mg of elemental calcium (but its absorption is much better).
Calcium citrate has about 21 percent elemental calcium.
However, if you suffer from acid stomach, it is probably better to avoid calcium citrate.
CALCIUM GLUCONATE
Another organic ingredient, calcium gluconate contains lower levels of calcium carbonate and calcium citrate. This type of calcium supplements are regarded as good but contain a very low amount of elemental calcium.
CALCIUM LACTATE
Combining calcium carbonate with lactic acid produces calcium lactate, an organic calcium in aged cheeses and baking powder. The ingredient features improved water solubility, and is used in functional beverages and as an antacid. Calcium lactate is regarded as good but contain a low amount of elemental calcium. Therefore, you will need to take a large amount of tablets to meet the calcium requirement.
CALCIUM ASCORBATE (FORM OF VITAMIN C)
You can get some calcium from a form of vitamin C called calcium ascorbate in which vitamin C is bound with calcium. Calcium ascorbate is regarded as an organic form of calcium, with the addition of vitamin C. Calcium ascorbate (calcium citrate) does not require stomach acid and therefore is also quite bioavailable.
CALCIUM PHOSPHATE AND TRIPHOSPHATE (TRICALCIUM PHOSPHATE)
Found in milk, calcium phosphate is less common in dietary supplements due to its low bioavailability. Calcium phosphate contains much lower percentage of elemental calcium. Calcium from calcium phosphate is not easily absorbed, except in the form of calcium triphosphate. Tricalcium phosphate, is closely related to the bone calcium but has a different crystal structure and is dehydroxylated, which results in lower acid requirements for dissolution and absorption. Calcium triphosphate has 39 percent elemental calcium.
CALCIUM CARBONATE
Calcium carbonate is an alkaline based chemical compound, with the chemical formula CaCO3. It is a common substance found as rocks in all parts of the world, and is the main component of shells of marine organisms and snails. It requires extra stomach acid for better absorption, so it is best taken between meals or not earlier than 30 minutes after meals, when stomach acidity starts getting lower again, or with a glass of acidic juice such as orange juice.
Carbonate provides more elemental calcium than calcium citrate (500 mg calcium carbonate provides 200 mg elemental calcium), so you may not need to take as many tablets or capsules, but it is an inorganic, harmful and much inferior form of calcium.
Calcium carbonate is a good antacid because it uses up stomach acid; if you are not producing excess stomach acid, calcium carbonate – such as oyster shells or limestone – is not the best choice.
Calcium carbonate is the active ingredient in agricultural lime, and is usually the key cause of hard water. It is commonly used medicinally as a calcium supplement or as an antacid. Calcium carbonate boasts approximately 40-percent elemental calcium, thereby using less space for a smaller pill. Calcium carbonate is found in rocks, marine animal shells and eggshells. Its alkaline base needs stomach acid for absorption. The ingredient offers low solubility, so it is not used in functional foods and beverages.
Calcium carbonate deposits have been found in the walls of major arteries in atherosclerosis (>).
Calcium carbonate with vitamin D results in elevated serum uric acid concentrations in elderly women (>, >).
Toxicity of calcium carbonate (>).
CALCIUM BICARBONATE
Calcium bicarbonate doesn’t occur in nature. It is formed when water containing carbon dioxide in solution (also known as carbonic acid) reacts with calcium carbonate. It forms in water when calcium carbonate and bicarbonate ions are dissolved with carbon dioxide.
DOLOMITE, BONE MEAL, OYSTER SHELL
These naturally occurring calcium supplements that are inorganic (high in calcium carbonate) and may contain heavy metals including mercury and lead. At the moment, calcium supplements are not tested by any regulatory agency for heavy metal content. Therefore, it’s best to avoid these.
CORAL CALCIUM
Those who sell supplements based on coral calcium sometimes claim that it can cure countless human health problems. In reality, however, it contains mostly inorganic calcium carbonate.
RECOMMANDED DAILY INTAKE
Adults under 50 need about 1000 mg of calcium a day, which amount may increase to 1,200 milligrams daily for adults over 50.
Children need between 200–700 milligrams per day depending on age, while teenagers should get about 1,300 mg per day to support their growing bones.
DOSAGE
If you need calcium supplements, I recommend 500 mg one to two times a day always with vitamin D3 (2000-5000 IU per day) and K2 (MK7 – 200-500 mcg per day) to prevent calcification and to improve calcium absorption.
It is not a good idea to take 1000 mg of supplements in one sitting as your body can only absorb 500 mg of calcium at a time. It’s best to split the 500 mg dose throughout the day.
Depending on age and gender, the daily calcium requirement ranges from 800 mg to 1300 mg for adults, so you need to do some calculations based on your calcium-rich food intake (one serving provides about 200-300mg).
INTERACTIONS WITH OTHER NUTRIENTS
Calcium and phosphorus have a see-saw relationship with each other; when one is high, the other is low. Excessive calcium intake, therefore, leads to phosphorus deficiency.
The best calcium sources should contain less phosphorus. Green leafy vegetables like kale, turnip greens, and mustard greens are excellent calcium sources as they have three to five times more calcium than phosphorus. Sesame seeds and tofu are good sources of both minerals as they have nearly equal amounts of calcium and phosphorus.
Beef, pork, and fish, on the other hand, have lots of phosphorus and very little calcium. Pepsi and carbonated beverages are loaded with sugar and typically may have 40-50 mg of phosphorus and no calcium.
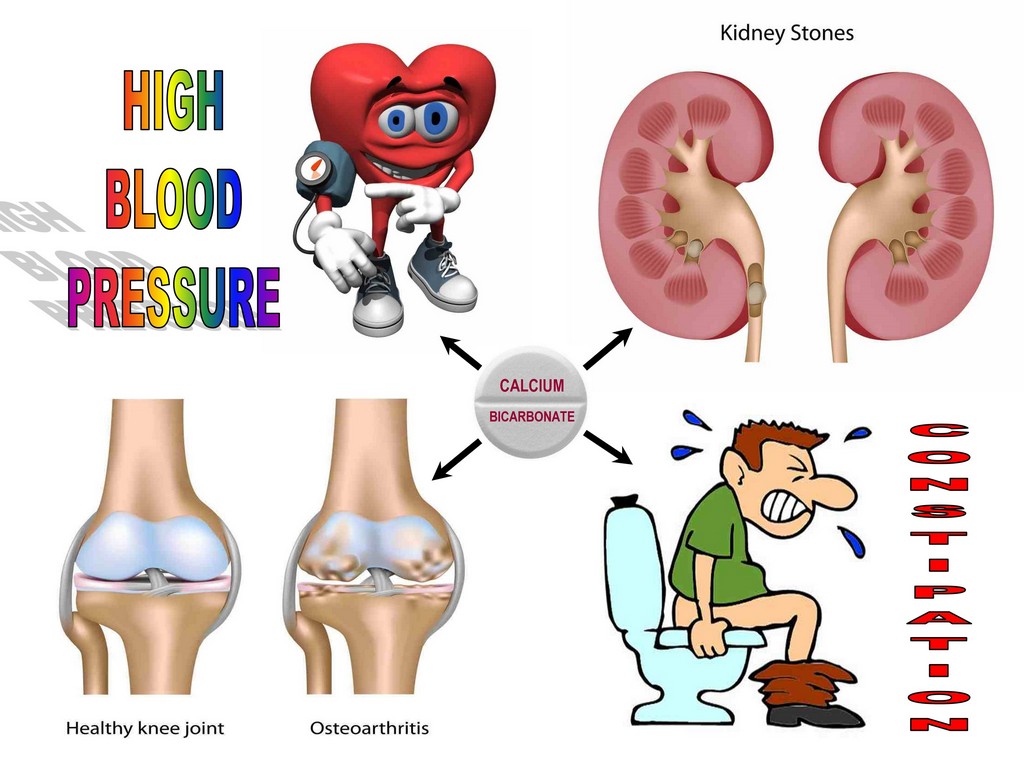
Excessive amounts of calcium lead to magnesium deficiency and increased risk of heart attack (heart is a muscle which requires magnesium), muscle cramps and spasms, neurological abnormalities, as well as other numerous problems (calcium causes muscle to contract while magnesium relaxes them).
Due to high dairy consumption and popular calcium supplementation Americans and Europeans tend to have a much higher calcium-to-magnesium ratio in their diet of about 3 to 1 or even worse.
In addition, people usually take calcium supplements high in cheap inorganic calcium carbonate (also present in hard water) which without magnesium, vitamin D, zinc, boron, and especially vitamin K2 accumulates in arteries and together with cholesterol hardens blood vessels leading to heart attacks and strokes.
CALCIUM CARBONATE AND PHOSPHATE MAY LEAD TO KIDNEY STONES
Taking a higher quantity of calcium carbonate and calcium phosphate supplements in order to increase the amount of elemental calcium intake can increase your risk of kidney stones and inhibit the absorption of iron and zinc.
There are four main types of kidney stones. An estimated 75 to 85 percent of all stones are made of calcium oxalate or calcium phosphate.
The next common type is called a struvite stone, which is made of magnesium, ammonium and phosphate.
There are also uric acid stones and cystine stones.
Calcium oxalate and calcium phosphate stones primarily form when people have a high amount of calcium in their urine. They can also develop from a kidney disease called renal tubular acidosis, from having a low urine level of a substance called citrate, or a high amount of oxalate or even vitamin C.

NEGATIVE EFFECTS OF EXCESSIVE CALCIUM INTAKE
There is still a lot of hype nowadays about getting more calcium to maintain strong bones. The recommended dietary allowance (RDA) for calcium in the US is 1,200 mg per day (in UK and EU it is 800 mg). As a result, Americans consume huge quantities of calcium (in the form of dairy and supplements) compared with most of the world. Therefore, we should assume that that they should have a very low risk of osteoporosis. In reality, however, the very opposite is true. The United States has one of the highest rates of osteoporosis in the world. It is interesting that the same is true with regards to Scandinavian countries where consumption of dairy (richest source of calcium) is highest in the world! Well, bot Americans and Scandinavians have weakest bones in the world in spite of highest calcium intake because of highest intake of dairy protein which steals calcium from their bones!
The best way to stop this calcium loss is avoid coffee and other sources of caffeine reduce consumption of sugar and especially animal protein (milk, dairy, cheese and also eggs and meat products), and be more physically active.
The dairy industry used media and health authorities to make people believe that we must get calcium from dairy products in order to maintain strong bones. It is very interesting that even in a study (sponsored by the Dairy Council) women consuming three 8-ounce glasses of cow’s milk per day lost calcium from their bones, and remained in negative calcium balance, even after a year of consuming almost 1,500 mg of calcium daily (in the form of milk)!
In order to prevent and reverse osteoporosis everything we need is a diet low in animal protein and high in a variety of unrefined plant foods especially raw vegetables, pulses (legumes), fresh fruits, whole grains and some seeds and nuts.
A 1981 study of calcium and protein levels showed that consuming 95 g of protein per day from animal sources and 1,400 mg of calcium supplements caused a net loss of nearly 30 mg of calcium from the bones every single day.
OTHER POSSIBLE NEGATIVE RESULTS OF TAKING CALCIUM SUPPLEMENTS
According to one study suggests excessive dietary calcium intake (from dairy) was associated with an increased prevalence of metabolic syndrome in overweight males (>).
There is also a strong relationship between the total calcium intake and an increased risk of cardiovascular disease mortality (>). The higher the intake of calcium the higher the risk, especially if you don’t take vitamin K2 (MK2 or MK4) supplements.
Increased calcium intake may also trigger constipation, diarrhea, and abdominal pain (>).
Drink eight cups of water throughout the day to avoid constipation.
CALCIUM IN HARD WATER

People today eat refined junk food and as a result they are deficient in magnesium or calcium but instead of improving their diet they try to get these minerals by drinking hard water. The problem, however, is that water is not supposed to nourish our body but cleanse it. It is the healthy unrefined diet which is to provide us with all essential nutrients, including minerals. These minerals found in healthy foods are organic and therefore well tolerated by the body and absorbed. Unfortunately, the minerals such as calcium carbonate, found in hard water, can’t nourish us as they are not organic and foreign to our body.
For this reason, the water you drink shouldn’t be hard (high in inorganic minerals) but as soft as possible (the lower in harmful inorganic minerals the better). The minerals your body need must be organic and they are found in unrefined healthy foods such as fresh vegetables, fresh fruits, whole grains, pulses, nuts and seeds. It means that water you drink every day must be as low in minerals as possible (soft water) because minerals in water are inorganic and are not beneficial but harmful. Especially dangerous is calcium carbonate as it contributes to kidney stones, hardening of arteries and heart attacks or strokes, arthritis and constipation. For this reason the best water to drink is distilled water as distillation not only removes all contaminants, such as heavy metals, hormones, antibiotics, dioxins, etc. but it is the only method which gets rid of inorganic minerals including harmful calcium carbonate.
What’s the difference between organic and inorganic calcium? It has to do with the natural life circle. The earth’s crust contains inorganic calcium, found in rocks. This inorganic calcium, magnesium and other minerals are absorbed by plants which are able to do what we can’t, they convert those inorganic minerals into organic ones our cells are able to absorb and use. For this reason in order to get beneficial organic calcium we are not supposed to drink water with inorganic calcium or consume chalk, limestone, oyster shell, egg shell, bone meal as it is dangerous and harmful. According to the statement by Greenmedinfo (based on scientific studies), „Growing evidence suggests that inorganic calcium ingestion in the form of Calcium Carbonate (chalk, limestone, oyster shell, egg shell, bone meal) is not a biologically appropriate form of calcium for human metabolism and is associated with calcification of soft tissue, osteoarthritis, constipation, kidney stones, hypertension and various other side effects of poorly utilized calcium.”
For the same reason we are to avoid water with inorganic minerals. And a simple conclusion is that since distilled water is completely free from harmful inorganic material it should be regarded as the best option. We can’t be benefited by calcium from rocks because the cells don’t recognize it as a viable life-giving element. If you feel you need calcium supplements try taking chelated calcium (such as citrate). Chelation makes calcium more organic by bounding calcium molecule to an amino-acid. As a result our cells can use it because they recognize it as a food. Calcium citrate seems to be the best example of chelated (organic) form of calcium supplement: „Calcium as found in chelated form, e.g. calcium citrate, or in its natural state as food, is much more readily absorbed and utilized in the body, and is safe.”
Apart from that, also calcium gluconate, aspartate or malate are regarded as good supplements. But please stay away from supplements containing inorganic calcium carbonate.
The best natural and organic form of calcium is of course the one found in food, especially in green leafy vegetables.
There is no doubt that the softer the water (the lower in inorganic harmful minerals) the better. Long time ago it was known that hard water was dangerous. For example, let me share with you a very interesting statement about soft and hard water written in 1866 by Ellen G White:

„Thousands have died for want of pure water and pure air. If they would accustom themselves to outdoor exercise, and to air in their houses, summer and winter, and use soft water for drinking and bathing purposes, they would be well and happy… On no single point is there need of enlightenment more than on this, of the superior quality of soft over hard water as a hygienic agent. Not only is hard water productive in many instances of diseased kidneys, irritation of the bladder, mucous dyspepsia, and scrofulous development, but as I have said before, its effects on the skin are to leave it rough, causing it to put on a dry, scaly appearance, making it to crack – and its effects on the mucous membrane are even worse, creating an irritation of that texture, serving to introduce dyspeptic conditions, sore throat, nasal catarrh, inactivity of the liver, costiveness, piles, and headache. Persons using it as a daily drink, never mingling it with anything else, would be marked over whole districts of country by habitual constipation, by dry skin, by shrivelled muscle, and are therefore, as if by instinct, led to avoid its use. I have known persons taking hygienic treatment for constipation of the bowels, whom physicians had utterly failed to cure by any hydropathic appliance, and have been compelled to resort to medicines, immediately relieved on the use of pure soft water as a drink. But this is only half its value. Its power as a solvent, as well as a tonic, its gentle and invigorating effect on free mucous surfaces, thus indirectly securing the health of all the senses, whose niceties of action depend on the health of the mucous tissue, are evidences of its advantage as a hygienic agent. The animals might teach us on this point. Horsemen, who study the natural conditions of their horses, and seek to preserve their health, are very particular in procuring soft water for them to drink, they having been taught that the taste of the horse is so perfect and nice in this matter, as to cause him even when thirsty, to refuse to drink at hard-water springs, and go for miles till he can find soft water. Besides, horsemen say that the remote effects of hard water as a drink for the horse, are, that instead of a glossy, sleek appearance of the hair which the horse shows when in the habit of drinking soft water, there arises a staring, dry, and apparently half-deadened condition of the hair and skin, making the grooming of the animal doubly difficult. I should advise every family who may read this lecture, and who use hard water either for drink or for culinary purposes, to take measures immediately to supply themselves with soft water in abundant quantities; and if it can only be obtained by being caught in reservoirs as it falls from the clouds, to filter it before it is used. Filtered rain-water is perfectly unobjectionable as a drink; and an expenditure of from five to twenty dollars in the purchase of a filter, would secure to any family in this land the means of purifying all the water they might need for drink and for cooking purposes.”

So, here we have a humble woman who lived in 19th century saying that soft water is much healthier to drink than hard water. But the whole medical world for decades have been teaching us that hard water is healthier because of the magnesium and calcium content. So, who is right? Long time I was trying to find any support for her claim and finally I found it in the book written by Dr James Balch, who in one of his books wrote: „Though some studies have shown that deaths from heart disease may be lower in areas where drinking water is hard, yet the calcium found in hard water is not good for the heart, arteries, or bones. Water deposits its calcium and other minerals on the outside of these structures, while it is the calcium and magnesium found within these structures that are beneficial to the body”. It means that calcium in hard water is harmful because it is deposited outside of bones causing calcification of blood vessels, joints, or pineal gland or contributing to kidney stones, haemorrhoids, varicose veins and other problems.
Since water is so contaminated today make sure you filter both tap and bottled water using at least inexpensive carbon (charcoal) water filters. Unfortunately, cheap carbon filters can remove only about 5 to may be 20 % of contaminants from water. So, if possible do your best to buy water distiller and drink distilled water (which is the best and most economical way to filter water) or if you still prefer filtered water you can install (under sink) a very effective reverse osmosis filter which includes carbon filter, reverse osmosis filter, alkaline filter and other elements which greatly improve the quality of tap water removing as much as 95 to 99% of contaminants found in water. Read more >
RELATED ARTICLES
SOURCES
– Weaver, C.M., Heany, R.P., Martin, B.R., Fitzsimons, M.L. 1991. Human calcium absorption from whole-wheat products. J. Nutr. 121:1769-1775.
– Howe, J.C. 1990. Postprandial response of calcium metabolism in postmenopausal women to meals varying in protein level/source. Metabolism. 39:1246-1252.
– Kitano, T., Esashi, T., Azami, S. 1988. Effect of protein intake on mineral (calcium, magnesium, and phosphorus) balance in Japanese males. J. Nutr.Sci.Vitaminol. 34:387-389.
– Kok, D.J., Iestra, J.A., Doorenbos, C.J., Papapoulos, S.E. 1990. The effect of dietary excesses in animal protein and sodium on the composition and the crystallization kinetics of calcium oxalate monohydrate in urines of healthy men. J.Clin.Endocrinol.Metab. 71:861-867.
– Greger, J.L., Krzykowski, C.E., Khazen, R.R., Krashok, C.L. 1987. Mineral utilization by rats fed various commercially available calcium supplements or milk. J.Nutr. 117:717.
– Bailie, I.E. 1987. Osteoporosis and ‘affluent diet’. Hospital practice. April 15, p16.
– Blank, R.P., Diehl, H.A., Ballard, G.T., Melendez, R.C. 1987. Calcium metabolism and osteoporotic ridge resorption: A protein connection. J.Prosthetet.Dent. 58:590-595.
– Specker, B.L., Vieira, N.E., O’Brie, K.O., Ho, M.L., Heubi, J.E., Abrams, S.A., Yrgey, A.L. 1994. Calcium kinetics in lactating women with low and high calcium intakes. Am.J.Clin.Nutr. 59:593-0.
– Pretice, A. Jarjou, L.M.A., Cole, T.J., Stirling, D.M., Dibba, B., Fairweather-Yait, S. 1996. Calcium requirements of lactating Gambian mothers: effects of a calcium supplement on breast milk calcium concentration, maternal bone mineral content, and urinary calcium excretion. Am.J.Clin.Nutr. 62:58-67.
– Johnson, Q. 1997. The effect of dairy consumption on reproductive health and other parameters of vervet monkeys. Ph.D. dissertation. (In prep)
– Campbell, Colin, M.D., China-Oxford Study
– RDA’s for Calcium, Nancy Clark’s Nutrition Cookbook. 1990.
– McDougall, John,M.D. Diet for A New America.
– Spencer, H. Do Protein and Phosphorus Cause Calcium Loss? Journal of Nutrition, 118:657.660; 1988
– Ellis, F. et al. Incidence of Osteoporosis in Vegetarian and Omnivores, American Journal of Clinical Nutrition, 25:555, 1972
– Hegsted, Relationship Between Nutrition in Early Life and Late Outcomes, Including Osteoporosis, Nutrition and Aging, 1990, pp.73-87
– Barnard, N.,M.D. Physicians Committee for Responsible Medicine, 1995.
– Hegsted, M Calcium and Osteoporsis, Journal of Nutrition, 116: 23162319; 1986
– Rogers, Sherry, MD: The Scientific Basis for Selected Environmental Medicine Techniques, 1994
– Spencer, H. Effect of a High Protein (Meat) Intake on Calcium metabolism, American Journal of Clinical Nutrition, 31:2167-2180; 1978.

Share this article!
Any information or product suggested on this website is not intended to diagnose, treat, cure or prevent any medical condition. Never disregard medical advice or delay in seeking it because of something you have read on this website. Consult your primary healthcare physician before using any supplements or making any changes to your regime.
© 2016 Slawomir Gromadzki – All Rights Reserved
